Previous work at BIRA-IASB and elsewhere has revealed the existence of an elusive but very large source of formic acid of unknown origin in the Earth’s atmosphere. Formic acid is the dominant acidifying agent in clouds in precipitation at remote locations.
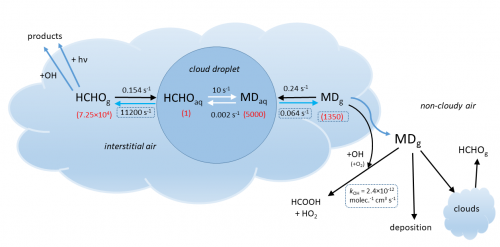
A few years ago, formaldehyde (HCHO) processing by liquid clouds was claimed to be a very large source of formic acid (HCOOH) explaining the missing source (Franco et al., Nature, 2021). This mechanism involves formaldehyde (HCHO) conversion into methanediol (HOCH2OH) in clouds and gas-phase oxidation of methanediol into formic acid.
Now, a new study just published in PNAS (Proceedings of the National Academy of Science) (Nguyen et al.) demonstrated using theoretical and modelling tools that this process accounts for less than 10% of the source required to close the global budget of formic acid. Moreover, the model simulations – performed at BIRA-IASB – show that the mechanism is especially inefficient over vegetated areas, which is precisely where the missing source is expected to be largest.
Theoretical assessment of key processes
Using advanced theoretical tools, Nguyen et al. found out that the gas-phase reaction of methanediol with OH is much slower (by a factor of 3) than assumed by Franco et al. on the basis of laboratory experiments. The discrepancy is explained by the presence of other compounds (oligomers of methanediol) contributing to HCOOH in the experiments, but not in the real atmosphere.
Next, high-level theoretical assessment of the equilibrium between gas-phase formaldehyde and methanediol in clouds shows that gas-phase methanediol is present in only tiny amounts (about 2% of the formaldehyde present), independently of the liquid water content of the cloud.
Simulating the impact of in-cloud formaldehyde processing on formic acid
Model simulations were performed at BIRA-IASB to quantify the conversion of aqueous methanediol to the gas phase when the cloud evaporates. The results were used to parameterize the overall cloud-processing mechanism in a global chemistry-transport model, the MAGRITTE model, which was then used to quantify the production of formic acid in the atmosphere through the cloud-processing mechanism.
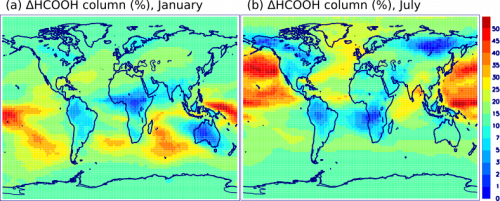
Great attention was paid to account for major sources of uncertainties, most notably the average time that air parcels spend in clouds. Other processes affecting methanediol besides its oxidation into formic acid – deposition on the Earth’s surface and re-incorporation in clouds – were found to be important for the final assessment. As shown on the Figure on the left, the relative impact of cloud-processing on formic acid is largest (up to +50%) over ocean – where clouds are abundant and other sources of formic acid are scarce – and it is lowest over forests.
It appears that the missing source of HCOOH remains decidedly elusive! More work and imagination will be needed to explore possible ways to resolve the longstanding discrepancy…
Publication
Nguyen, T.L .; Peeters, J .; Müller, J.-F .; Perera, A .; Bross, D.H .; Ruscic, B .; Stanton, J.F. (2023). Methanediol from cloud-processed formaldehyde is only a minor source of atmospheric formic acid. , Proceedings of the National Academy of Sciences, Vol. 120, Issue 48, e2304650120, DOI: 10.1073/pnas.2304650120.
Scientific contact
- Dr. Jean-François Müller, Head of Department "Sources and sinks of atmospheric constituents", Royal Belgian Institute for Space Aeronomy
E-mail : jean-francois (dot) muller (at) aeronomie (dot) be
Press contact
- Dr. Karolien Lefever, Head of “Communication and documentation”, Royal Belgian Institute for Space Aeronomy
E-mail: karolien (dot) lefever (at) aeronomie (dot) be